 |
| Structural origin of the liquid-liquid transition. © 2015 Ken-ichiro Murata, Hajime Tanaka |
A University of Tokyo research group has successfully identified a microstructural unit that controls liquid-liquid transition between two phases in a single substance with multiple liquid phases. Identifying this unit is key to understanding liquid-liquid transitions.
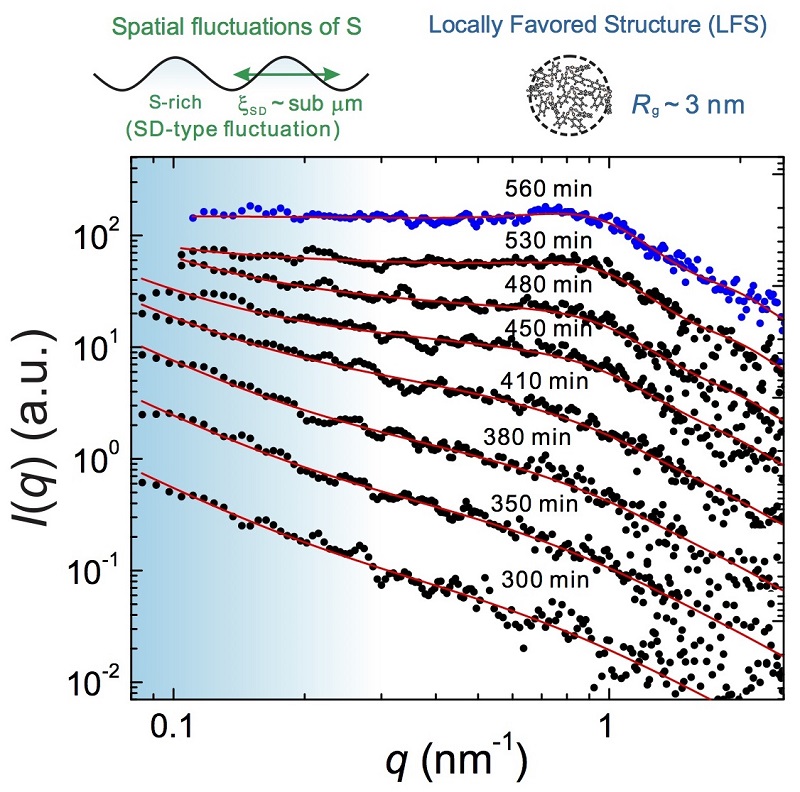
It is widely known that even a single-component substance can have more than two crystals, as in the case of carbon (diamond and graphene) and water. Contrarily, it was thought that as a liquid is a disordered state there is only one liquid state for a single-component substance. Liquid-liquid transition in such single-component substances has attracted considerable attention as a new type of phase transition, overturning the conventional view of liquids. However, although much evidence suggestive of its presence has been gathered, the existence of liquid-liquid transitions is still an ongoing debate due to experimental difficulties. To prove the existence of liquid-liquid transitions, it is necessary to experimentally identify the micro structure governing liquid-liquid transition on a microscopic level.
Professor Hajime Tanaka’s research group at the Institute of Industrial Science have successfully identified a structural unit that controls a liquid-liquid transition by using an organic liquid, triphenyl phosphite, which has a transition under ambient pressure. The research group observed the target liquid by irradiating it with X-rays and found that the new liquid formed after the transformation has a higher density of clusters composed of several molecules.
Professor Tanaka says “A liquid state is one of the fundamental states of matter besides gas and solid, and an important physical state universal to a wide range of materials including metals, semiconductors, and organic materials. Thus, our finding not only contributes to our understanding of the underlying mechanism of liquid-liquid transition, but also provides a new insight into the liquid phase, which has been believed to be uniform and random, and leads to a deeper understanding of the very nature of the liquid state.”
Paper
, "Microscopic identification of the order parameter governing liquid-liquid transition in a molecular liquid", Proceeding of the National Academy of Sciences of the United States of America Online Edition: 2015/4/27 (Japan time), doi: 10.1073/pnas.1501149112.

No comments:
Post a Comment